Comparative Healing Efficacy of Platelet Rich Fibrin and Zinc Oxide Nanoparticles on Critical-Sized Ulnar Bone Defects in Rabbits
Abstract
Objective
Evaluation of the ability of autogenous Platelet Rich Fibrin (PRF) and Zinc Oxide Nanoparticles (ZnONPs) to repair critical-sized ulnar defects in rabbits based on radiographic assessment.
Design
Randomized controlled study.
Animals
Twenty-four healthy male white New Zealand rabbits with an age of 6.0 ± 0.3 months and weight of 2.5 ± 0.29 Kg were used.
Procedures
A 12 mm defect was created in the diaphysis of the right ulnae in allrabbits,then the rabbits were randomly allocated into three groups (8 each): Control group, (the defect left for healing without grafts), platelets rich fibrin group (PRF group, the defect filled with PRF) and combination group (PRF/ZnONPs group, the defect filled with both PRF and ZnONPs). Healing capacity between the groups was evaluated by immediate postoperative radiographic assessment and subsequently at the first and the second postoperative months.
Results
Statistical analysis showed significant differences in the radiographic healing score between the groups (P = 0.000) at all-time points (P = 0.000- 0.003) during the study. Rabbits in the combination group showed the highest radiographic healing scores followed by the PRF group meanwhile, the Control group showed minimal radiographic healing scores.
Conclusion and Clinical Relevance
The addition of ZnONPs to PRF can accelerate the healing of ulnar critical-size defects in rabbits.
Author Contributions
Academic Editor: Mohammed A Elmetwally, Animal Science Department Texas A&M University, USA 442D Kleberg Animal and Food Sciences Center 2471 TAMU College Station, TX 77843-2471
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2021 Esraa Zalama, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Repair of segmental long bone defects, especially the true CSD1 represents one of the most challenging situations in orthopedic surgery. Such a defect most probably requires surgical intervention as it is not able to heal spontaneously1, 2. Open fractures, osteoclastic tumors, and gunshot wounds are most of the clinical incidents leading to primary bone defects while the secondary bone defects involve a surgical bone resection that occurs mainly in some clinical situations such as bone tumors, infections, and non-unions 3.
Note:
1Critical sized bone defects
Bone regeneration can be achieved by bone tissue engineering via osteoinductive growth factors, osteoconductive scaffolds, and osteogenic cells, or their combination which has been established as an alternative technology to substitute conventional autograft or allograft treatments and to provide an appropriate environment for osteoinduction in large bone defects 4, 5
PRF is a second-generation platelet concentrate 6 first described by Choukroun et al. (2001) 7 and inspected as an alternative method for better bone regeneration8. The fibrin matrix of PRF is obtained as a result of slow polymerization and it can hold many growth factors and cytokines, and prolong the time of their release in the wound site 9. It also can trap the circulating stem cells which act as a natural guide for angiogenesis and provides wound protection by epithelial coverage 10, 11, 12. PRF is rich in fibrin; leukocytes; cytokines; circulating stem cells; vascular endothelial growth factor; insulin-like growth factor-I and II; and platelet-derived growth factor 6 which are shown to accelerate bone repair, increase the rate of collagen formation and promote fibroblast and osteoblast proliferation 13. Several authors have also demonstrated that optimal support for mesenchymal stem cells is mainly provided by the fibrin matrix 10, 14, which contributes to the regeneration of osseous defects and many other tissues.
NPs2 are modified particles at the atomic level (1–100 nm) possessing special properties and used in a variety of novel applications 15 . ZnONPs are one of the metal oxides used in tissue repair, the food industry, and in cosmetics such as lotions and sunscreens as it is known to possess UV absorbing and blocking properties16. They also have been widely used in numerous potential applications in veterinary sciences due to their unique properties such as antimicrobial and antitumor activities, wound healing, and angiogenic ability 17. Applications of ZnONPs in veterinary medicine include the treatment of meningitis, tumors, and diseases caused by intracellular pathogens like viruses, bacteria, and fungi where it is really difficult for the macromolecular therapeutic substances to access the target site or cross the different body barriers, unlike the nanoparticles, due to their minute size and high surface area 18.
Note:
2Nanoparticles
Finally, the osteogenic properties of ZnONPs and their ability to enhance osteoblast adhesion and growth were also considered by Foroutan et al. 19 who reported that administration of ZnONPs actively contributed to the osteogenic differentiation of bone marrow mesenchymal stem cells into osteoblasts. Fabrication of ZnONPs-containing scaffolds for bone defect regeneration is the main use of ZnONPs in bone tissue engineering20.
The main hypothesis of this study is that the use of PRF as a platelet concentrate scaffold in addition to ZnONPs will provide sufficient growth factors and stimulators for the critical bone defect healing which may reduce the healing time and the animal will have a quiet recovery. Thus, the present study was designed to evaluate the osteogenic capacity of both PRF and ZnONPs in the repair of critical size ulnar defects in rabbits.
Materials and Methods
Animals and Experimental Design
Twenty-four male New Zealand white rabbits with an age of 6.0 ± 0.3 months and weight of 2.5 ± 0.29 Kg were used. A 12 mm defect was created in the diaphysis of the ulnae in one limb while the contralateral limb was kept intact in all experimental rabbits. Then the rabbits were classified into three experimental groups (n=8): control group; (no materials were added), platelets rich fibrin group (PRF group); defects were filled with autogenous PRF) and combination group (PRF/ZnONPs group); defects were filled with both PRF and ZnONPs). This study was approved by the Ethics Committee of the Faculty of Veterinary Medicine, Mansoura University, Egypt, and registration number (M/6).
Preparation of Autogenous PRF Graft
Autogenous PRF was prepared from each rabbit as mentioned by Choukroun et al. 7 technique, where 3 mL of whole blood was obtained after aseptic preparation from the ear central or lateral veins of each rabbit in a 4 ml plain tube thereafter the blood was centrifuged immediately at a rate of 3000 rpm for 10 min till separation into three distinct layers. The fibrin clot in the middle layer (Figure 1), is considered the leukocyte-rich PRF which is carefully taken by non-toothed tissue forceps and separated from the RBCs layer with avoidance of the PRF membrane rupture.
Figure 1.Preparation of autogenous PRF. A: The blood in a plain tube was separated into three distinct layers where the middle layer, a strong fibrin clot, represents the leukocyterich PRF. B: A clean yellowish white PRF clot was obtained after whipping the adhered RBCs layer off on a sterile gauze pad.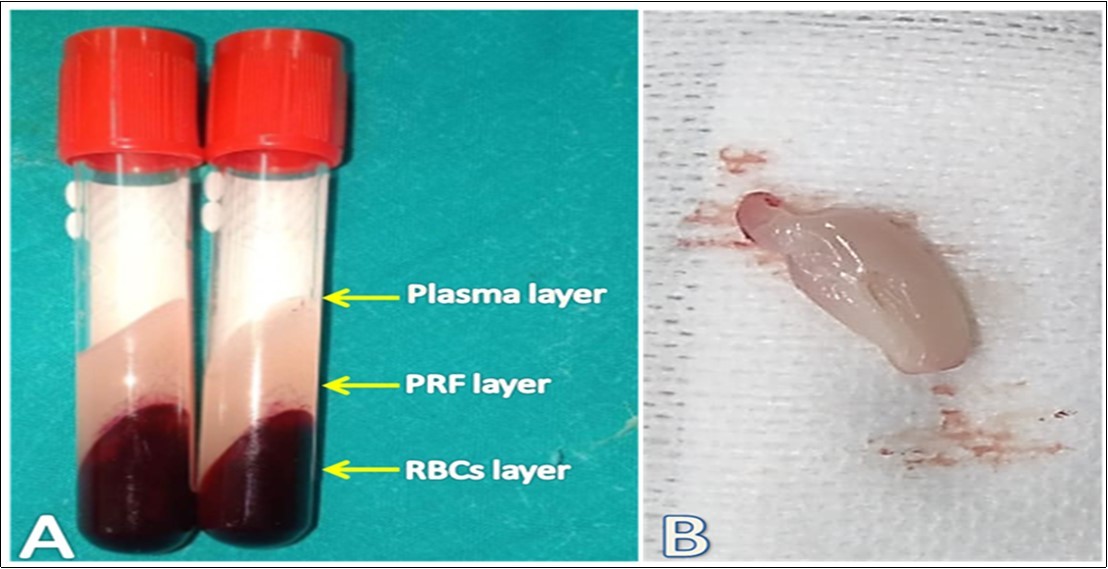
Preparation of Zinc Oxide Nanoparticles
In this study, ZnONPs, milky dispersion (Sigma-Aldrich chemicals, Nasr City, Cairo, Egypt) with particles size of <100 nm, the concentration of 20 wt. % (in H2O), the density of 1.7 ±0.1 g/mL (at 25 °C) and pH of 7.5±1.5 were purchased and kept in a dark place at room temperature. The dispersion was diluted to 0.2% using 0.9% normal saline. In the PRF/ZnONPs group, 0.1 ml of ZnONPs 2% (ZnONPs density of 1.72± 0.1 mg) was injected carefully into the PRF clot membrane using an insulin syringe. The ZnONPs dose was selected according to our unpublished pilot study performed before this study.
Surgical Procedures
General anesthesia was achieved using an IM3 injection of a combination of 7.5 mg kg-1 xylazine (20 mg/ml; Xylaject; ADWIA Co, Egypt) and 35 mg kg-1 ketamine hydrochloride (Ketamine 50 mg /10 ml, Rotexmedica, Germany). On lateral recumbency, the ulnar shaft is approached by a craniolateral incision in the antebrachial region. Exposing the right ulna was done by incising the antebrachial fascia between the ulnaris lateralis muscle and the lateral digital extensor muscle and retracting the muscles. Then, a 12-mm defect was osteotomized from the shaft of the ulna using a low-speed electric drill (APT, China) with a 1.5 ml diameter drill bit. In the control group, the defect was left empty. While in the PRF group, a PRF clot was used to fill up the defects. In the PRF/ZnONPs group, 0.1 ml of ZnONPs dispersion 2% (2 mg ZnONPs) was inoculated carefully into the PRF clot membrane using an insulin syringe. The subcutis and the skin were closed by the routine manner using 3-0 vicryl (ETHICON, USA) and 3-0 silk (ETHICON, USA), respectively. A protective bandage was applied and then all rabbits were allowed to recover in an incubator warmed to 37-40 °C until fully awake. Afterward, they were moved to their cage. All animals received meloxicam (meloxicam 15 mg, AMRIYA pharmaceutical company, Egypt) at a dose rate of 0.6 mg/kg for 7 days. The antibiotic regimen included Cefotaxime sodium (Cefotaxime sodium, EIPICO pharmaceutical company, Egypt) at a dose rate of 50 mg/kg bodyweight for 5 days. The incisions were dressed daily with povidone-iodine and bivatracin topical antibiotic spray (Neomycin sulfate 165000 IU, Bacitracin Zinc 12500 IU, and Aerosol Hydrocarbon to 150 ml, ECAP, Egypt). The feeding condition, weight, body temperature, and breathing were investigated daily.
Note:
3Intramuscular
Radiographic Examination
Standardized mediolateral and anteroposterior radiographs were performed immediately after the operation and subsequently on the 1st and the 2nd postoperative months at 40 kV, 100 mA, 10 mAs, and 80 cm FFD4 using X-ray machine, Poskom Co., Korea. General anesthesia of rabbits was performed before radiological examination by administration of a combination of 5 mg kg-1 xylazine (20 mg/ml; Xylaject) and 35 mg kg-1 ketamine hydrochloride (Ketamine 50 mg /10 ml), injected IM in one syringe.
Note:
4Focal Film Distance
Compared to the immediate postoperative radiographs, healing of the defect was determined by evaluating the radiographs in each group at the 1st and the 2nd postoperative months for bridging the gap, and the healing capacity of each treatment. The healing score was measured by different independent operators using a ruler to measure the percentage of newly formed tissue in the defect depending on the apparent greyscale. Our healing score was designed as follows, score 1 = less than 25% healing, score 2 = 25-50% healing, score 3 = 50-75% healing, and score 4=75-100% healing. Bridging of the gap is considered present if any newly formed growth was found to connect between the two extremities of the defect on X-ray films. Otherwise, gap bridging is considered absent. The higher healing score with gap bridging was considered to have more new bone formation.
Statistical Analysis
Statistical analyses were performed according to Elsaied et al. 2020 and Samy et al. 2020a, b 21, 22, 23 using SPSS (version 25.0, Inc, USA). Gap bridging results were expressed as frequency while healing scores were expressed as median (minimum-maximum). To determine the effect of time, and group on the healing score, Kruskal-Wallis nonparametric ANOVA test was used meanwhile chi-square was used with gap bridging frequencies. For all analyses, P ≤ 0.05 was considered significant.
Results
During the current study, no serious complications and death were observed and the healing took place in all surgical wounds by the first intention. Clinical wellness was observed in all rabbits as early as the second postoperative day. Compared to the postoperative radiographs, the addition of PRF and ZnONPs to segmental ulnar CSD of 12-mm length resulted in obvious radiographic changes (Table 1, Figure 2), and the statistical analysis showed a significant difference in the healing score between the three experimental groups (P = 0.000) at all-time points (P = 0.000- 0.003) during the study.
Table 1. Results of statistical analyses.| Healing score | Group | 0 day | 30th day | 60th day | Kruskal-Wallis H | P value |
| Control | 1.0(1.0-1.0)b | 2.0(1.0-2.0)a* | 2.0(2.0-2.0)a* | 11.9 | 0.003 | |
| PRF | 1.0(1.0-1.0)c | 3.0(2.0-3.0)b** | 4.0(3.0-4.0)a** | 15.63 | 0 | |
| PRF/ZnONPs | 1.0(1.0-1.0)b | 4.0(4.0-4.0)a*** | 4.0(4.0-4.0)a** | 17 | 0 | |
| Kruskal-Wallis H | 0 | 15.59 | 15.57 | |||
| P value | 1 | 0 | 0 | |||
| Bridging callus | Group | 0 day | 30th day | 60th day | Fisher's Exact test | P value |
| Control | 0% | 0% | 75% | 13.568 | 0.001 | |
| PRF | 0% | 62.50% | 100% | 17.46 | 0 | |
| PRF/ZnONPs | 0% | 62.50% | 100% | 17.46 | 0 | |
| Fisher's Exact test | 0 | 8.97 | 2.98 | |||
| P value | 1 | 0.018 | 0.304 |
Figure 2.Showing lateral radiographs of the three experimental groups (Control, PRF and PRF/ZnONPs) at 0, 30 and 60 postoperative days.
Concerning bone healing in the region of interest, treatment of CSD by adding PRF either alone (PRF group) or in combination with ZnONPs (PRF/ZnONPs group) resulted in a highly significant (P = 0.000) increase in the healing score of CSD versus the Control group. On the 30th postoperative day, the healing score was 3.0 (2.0- 3.0) and 4.0 (4.0- 4.0) in PRF and PRF/ZnONPs groups respectively versus 2.0 (1.0- 2.0) in the Control group. On the 60th postoperative day, complete filling of the CSD represented by 0.4 healing scores was observed in both treatment groups, versus 2.0 healing score and incomplete filling in the Control group.
In terms of bridging callus, there was a significant difference between times within a group (P = 0.000- 0.001) but a non-significant difference between groups within time. On the 30th day, 62.5% of rabbits in both treatment groups showed gap bridging versus no bridging in the control group with a significant difference (P = 0.018) between groups within this time point. At the end of the study, 75% of the control group rabbits started to show some sort of bridging callus meanwhile all rabbits of both PRF and PRF/ZnONPs groups exhibited complete bridging of the gap.
Comparing the radiographic density of the healed bone, both treatment groups exhibited higher bone density compared to the Control group. In all rabbits of the combination group, maximal density was early obtained on the 30th postoperative day.
Discussion
Regarding long bone CSD, diaphysis is the most common site of involvement.69% of all fractures associated with bone loss reported in the Edinburgh Trauma Unit were involving the diaphysis, with the rest having either loss of metaphyseal bone or the articular surface, or both 1. The rabbit ulna or radius defect (between 10 and 20-mm) is often used in scientific research as it is of relatively low cost and does not require any fixation owing to the support offered by the adjacent bone due to the fibro-osseous connection between these two bones 24. This is why the ulnar diaphyseal segmental CSD without internal fixation is decided to be used and evaluated as a surgical model in this study.
PRF is a fibrin clot in which cytokines, platelets and cells are trapped and then released slowly within a certain time 25. The use of PRF in clinical field includes many advantages such as a simple preparation technique without the need for external biochemical additives to the blood. This makes this preparation strictly autogenous which prevents tissue reactions or rejection. Additionally, it provides more tensile strength and adhesiveness properties for clot stabilization. Therefore, using it as an autologous scaffold for periosteal cells and osteoblasts has shown to be able to promote higher cell proliferation and metabolic activity 10, 26
Most studies involved the use of PRF for soft tissue repair, while the use of its fibrin-based material solely as a filling material for large bone defects remains controversial 27. Positive results were reported by Ozdemir et al. 28 for using PRF in combination with titanium barriers including increased newly formed bone quality and enhanced bone formation rate, attributed to the concentration of growth factors. In a study by Singh et al. 29, PRF was shown to promote bone and soft tissue healing in 40 surgical areas in humans. Kim et al. 30 also reported increased bone mineral density and bone volume after 6 weeks in calvaria bone defects of rabbits that were treated with PRF.
PRF has been solely used as a filling for the ulnar segmental CSD in this experiment without the addition of any bone substitutes as the fibrin matrix can be considered a scaffold. Good filling of the defect appeared in the radiographs as early as the first month and then filling continued till the end of the study where the defect was completely bridged and filled with newly formed bone and the highest healing score was achieved by the end of the second month. The low density of the bridging bone was noticed at the early stage of healing but then it becomes denser overtime.
In-vitro investigation of the possibility to improve the degradation and mechanical properties of TCP5 scaffolds by the addition of ZnONPs has been successfully done by Feng et al. 31. An optimal amount of ZnONPs (2.5 wt. %) was found to highly improve mechanical properties (compressive strength and fracture toughness) and bioactivity of the scaffolds. Once cultured on the surface of the composite scaffolds, The attachment and proliferation ability of osteoblast-like human cells were better than for the control sample. Moreover, after incubation of the optimized scaffolds in SBF6 for twenty-eight days, a layer resembling bone apatite was successfully obtained. Zn2+ ions are well-known to stimulate bone formation and mineralization and to have an active role in the proliferation of osteoblastic cells. These properties were contributed to the observed osteoconduction (guiding bone growth) and osteoinduction (formation of new bone tissue) which is assumed to be achieved by the released Zn ions from the TCP-ZnO composite scaffold 32, 33
Note:
5Tri-calcium phosphate
6Simulated body fluid
Similar findings were reported also by Fielding et al. 34 , who designed a novel 3D-printed TCP scaffold incorporating 0.25 wt. % ZnONPs and confirmed the boosting effects of Zn ions towards osteoblast cells. In addition, these findings were further demonstrated in vivo from the same research group 35 where the scaffolds were completely infiltrated by the newly formed bony tissue after three months postoperatively.
Similarly, the findings of this study support the results of the studies mentioned above. Our scaffold was a PRF incorporating 0.2% ZnONPs. Inoculating the specifically used dose of ZnONPs into the PRF matrix enclosed by its membrane has lowered the rate of Zinc ions release along with the release of growth factors naturally comprising the PRF fibrin matrix and this prolonged the ZnONPs osteogenic action. Statistical analyses showed a significant increase in the healing score in the PRF/ZnONPs group over the other two groups. Also, the use of ZnONPs has accomplished the highest bone density along with the study which proves that it enhances the PRF mineralization capacity and thus the newly formed bone density.
Conclusion
The results of the present study indicated that the addition of ZnONPs to the PRF can improve the bone defect healing criteria based on radiographic assessment.
Authors’ Contribution to the Study
Esraa Zalama: Experimental design, surgical procedures, animals follow up, statistical analysis and manuscript writing.
Alaa Samy: Study protocol & experimental design, surgical procedures, statistical analysis, editing & reviewing.
Awad Rizk: Study protocol & experimental design, surgical procedures, editing & reviewing.
Basma Salama: Conceptualization, experimental design, writing of the manuscript, providing ZnONPs.
Gamal Karrouf: Supervising the study & experimental design, editing & reviewing.
References
- 1.J F Keating, Simpson A H R W, C M Robinson. (2005) The management of fractures with bone loss. , J. Bone Joint Surg. Br 87, 142-150.
- 2.H C Fayaz, P V Giannoudis, M S Vrahas, R M Smith, Moran C et al. (2011) The role of stem cells in fracture healing and nonunion. , Int. Orthop 35, 1587-1597.
- 3.Wiese A, Pape H C. (2010) Bone defects caused by high-energy injuries, bone loss, infected nonunions, and nonunions. , Orthop. Clin. North Am 41, 1-4.
- 4.Petite H, Viateau V, Bensaïd W, Meunier A, C de Pollak et al. (2000) Tissue-engineered bone regeneration. , Nat. Biotechnol 18, 959-963.
- 5.A J Salgado, O P Coutinho, R L. (2004) Bone tissue engineering: state of the art and future trends. , Macromol. Biosci 4, 743-765.
- 6.D M, Choukroun J, Diss A, S L Dohan, Dohan A J J et al. (2006) Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part I: technological concepts and evolution. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. , Endod 101, 37-44.
- 7.Choukroun J, Adda F, Schoeffler C, Vervelle A. (2001) Une opportunit?? en paro-implantologie: Le PRF.Implantodontie.
- 8.Fabbro Del, Corbella M, Taschieri S, Francetti S, L et al. (2014) Autologous platelet concentrate for post-extraction socket healing: a systematic review. , Eur. J. Oral Implantol 7, 333-344.
- 9.Ehrenfest Dohan, Peppo D M de, G M Doglioli, P, Sammartino G. (2009) Slow release of growth factors and thrombospondin-1 in Choukroun’s platelet-rich fibrin (PRF): a gold standard to achieve for all surgical platelet concentrates technologies. , Growth Factors 27, 63-69.
- 10.Gassling V, Douglas T, P H Warnke, Açil Y, Wiltfang J et al. (2010) Platelet-rich fibrin membranes as scaffolds for periosteal tissue engineering. Clin. Oral Implants Res. 21, 543-549.
- 11.A C Brown, T H Barker. (2014) Fibrin-based biomaterials: modulation of macroscopic properties through rational design at the molecular level. , Acta Biomater 10, 1502-1514.
- 12.Kawase T, Kamiya M, Kobayashi M, Tanaka T, Okuda K et al. (2015) The heat-compression technique for the conversion of platelet-rich fibrin preparation to a barrier membrane with a reduced rate of biodegradation. , J. Biomed. Mater. Res. Part B Appl. Biomater 103, 825-831.
- 13.R V Kumar, Shubhashini N. (2013) Platelet rich fibrin: a new paradigm in periodontal regeneration. , Cell Tissue Bank 14, 453-463.
- 14.Ehrenfest Dohan, D M, P de, M Del Corso G, M et al. (2010) Choukroun’s platelet-rich fibrin (PRF) stimulates in vitro proliferation and differentiation of human oral bone mesenchymal stem cell in a dose-dependent way. , Arch. Oral Biol 55, 185-194.
- 15.Buzea C, I, Robbie K. (2007) Nanomaterials and nanoparticles: sources and toxicity. , Biointerphases 2, 17-71.
- 16.Becheri A, Dürr M, Nostro Lo, P, Baglioni P. (2008) Synthesis and characterization of zinc oxide nanoparticles: application to textiles as UV-absorbers. , J. Nanopart. Res 10, 679-689.
- 17.etchthanasombat C, Tiensing T, Sunintaboon P. (2012) Synthesis of zinc oxide-encapsulated poly(methyl methacrylate)-chitosan core-shell hybrid particles and their electrochemical property. , J. Colloid Interface Sci 369, 52-57.
- 18.Manuja A, Kumar B, R K Singh. (2012) Nanotechnology developments: opportunities for animal health and production. , Nanotechnol. Dev 2, 4.
- 19.Foroutan T, Mousavi S. (2014) The effects of zinc oxide nanoparticles on differentiation of human mesenchymal stem cells to osteoblast. , Nanomedicine Journal
- 20.Laurenti M, Cauda V. (2017) Zno nanostructures for tissue engineering applications. Nanomaterials (Basel). 7.
- 21.Elsaied N, Samy A, Mosbah E, Zaghloul A. (2020) Induction of Surgical Obstructive Cholestasis in rats: morphological, biochemical and immunohistochemical changes. , Mans. Vet. Med. J 21, 107-115.
- 22.Samy A, El-Adl M, Rezk S, Marghani B, Eldomany W et al. (2020) The potential protective and therapeutic effects of platelet-rich plasma on ischemia/reperfusion injury following experimental torsion/detorsion of testis in the Albino rat model. Life Sci. 256, 117982.
- 23.Samy A, Elmetwally M, S A El-khodery. (2020) Anti-nociceptive effect of intravenous regional analgesia in horses underwent selected short time distal limb surgeries. , J. Equine Vet. Sci 103113.
- 24.E A Horner, Kirkham J, Wood D, Curran S, Smith M et al. (2010) Long bone defect models for tissue engineering applications: criteria for choice. Tissue Eng. , Part B Rev 16, 263-271.
- 25.Mosesson M W. (2005) Fibrinogen and fibrin structure and functions. , J. Thromb. Haemost 3, 1894-1904.
- 26.Gassling V, Purcz N, Braesen J-H, Will M, Gierloff M et al. (2013) Comparison of two different absorbable membranes for the coverage of lateral osteotomy sites in maxillary sinus augmentation: a preliminary study. , J Craniomaxillofac Surg 41, 76-82.
- 27.Lee J-W, Kim S-G, Kim J-Y, Lee Y-C, Choi J-Y et al. (2012) Restoration of a peri-implant defect by platelet-rich fibrin. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 113, 459-463.
- 28.Ozdemir H, Ezirganli S, Kara Isa, Mihmanli M, A et al. (2013) Effects of platelet rich fibrin alone used with rigid titanium barrier. , Arch. Oral Biol 58, 537-544.
- 29.Singh A, Kohli M, Gupta N. (2012) Platelet rich fibrin: a novel approach for osseous regeneration. , J. Maxillofac. Oral Surg 11, 430-434.
- 30.Kim T-H, Kim S-H, G K Sándor, Kim Y-D. (2014) Comparison of platelet-rich plasma (PRP), platelet-rich fibrin (PRF), and concentrated growth factor (CGF) in rabbit-skull defect healing. , Arch. Oral Biol 59, 550-558.
- 31.Feng P, Wei P, Shuai C, Peng S. (2014) Characterization of mechanical and biological properties of 3-D scaffolds reinforced with zinc oxide for bone tissue engineering. , PLoS ONE 9 87755.
- 32.Hashizume M, Yamaguchi M. (1993) Stimulatory effect of beta-alanyl-L-histidinato zinc on cell proliferation is dependent on protein synthesis in osteoblastic MC3T3-E1 cells. , Mol. Cell. Biochem 122, 59-64.
- 33.Wei X, Akinc M. (2005) Si, Zn-Modified Tricalcium Phosphates: A Phase Composition and Crystal Structure Study. Key Eng. Mater.284-286: 83-88.
Cited by (2)
This article has been cited by 2 scholarly works according to:
Citing Articles:
Frontiers in Bioengineering and Biotechnology (2024) Crossref
Lauma Ieviņa, A. Dubnika - Frontiers in Bioengineering and Biotechnology (2024) Semantic Scholar
Frontiers in Bioengineering and Biotechnology (2024) OpenAlex
